Part 1:
Explanation: In this lab we were testing osmosis and diffusion. We were testing if diffusion were to occur what would happen. Would the starch diffuse through the dialysis bag or would the starch. Or would both.
Hypothesis:If diffusion occurs during the experiment then there will be glucose in the water of the cup because of diffusion from a high to a low concentration.
Materials:
Explanation: In this lab we were testing osmosis and diffusion. We were testing if diffusion were to occur what would happen. Would the starch diffuse through the dialysis bag or would the starch. Or would both.
Hypothesis:If diffusion occurs during the experiment then there will be glucose in the water of the cup because of diffusion from a high to a low concentration.
Materials:
- Goggles
- 1 plastic cup
- 15 cm Soaked dialysis tubing
- 15 mL 15% Glucose/ 1% Starch solution in a medicine cup
- 1 plastic pipet
- 2 glucose test strips
- 2 10 m pieces of string
- iodine(starch indicator) solution
- Fill cup up with distilled water. Leave about 1-3 cm off the top of the cup
- Dip glucose test strip in water, 1-2 seconds. Remove any excess liquid
- Wait 2-3 minutes to observe any color change on the strip. A positive (+) glucose test is indicated by a greenish color on the test strip. No color change will occur if the test result is negative (-). Record on Data table 1
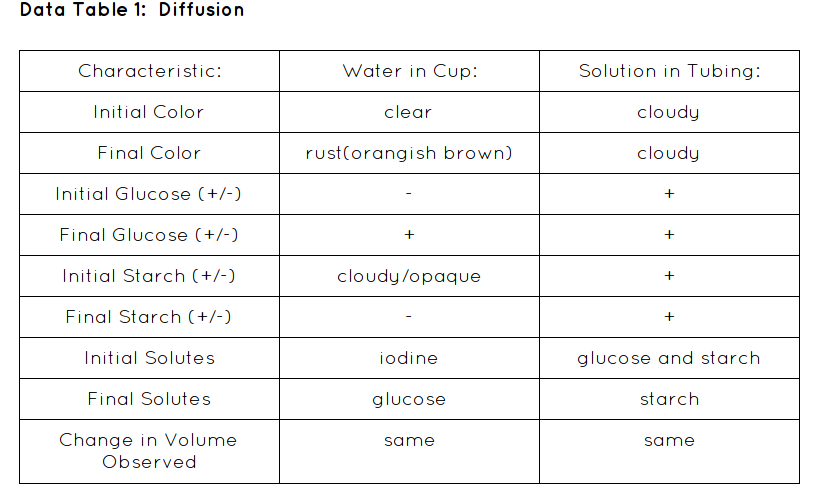
- Use a plastic pipet add 20-25 drops of the starch indicator solution (Iodine) to the water in the cup. Observe (take a picture) as it mixes with the water. Record the color of the water in Data table 1.
- Gently rub the tubing between your fingers to open it.
- Tie one end of the tubing tightly with a piece of string. Fill the tubing with water and test it for leaks at a sink then empty the tubing.
- Insert the funnel tip into the open end of the dialysis bag, and pour the 15 mL of 15% glucose/1% starch solution from the medicine cup into the tubing.
- Squeeze all the air bubbles out of the tubing and tie the open end shut with another piece of string. Note the color of the starch-glucose solution in the dialysis tubing and record in Data table 1.
- Rinse the outside of the bag under running water. Squeeze the bag gently to ensure that there are no leaks. Adjust the string if there are leaks.
- Completely submerge the model cell into the cup of water and starch indicator solution. Allow osmosis and diffusion to occur for 30 min.
- After 30 min test the water in the cup for sugar content as in Step 2. Note any color changes in the dialysis tubing and in the cup (take a picture). Record in Data table 1.
- Wash your hands and clean up your area.
Data Table:
Conclusion:
In our lab it went pretty well. We saw diffusion in action. According to my hypothesis was correct; If diffusion occurs during the experiment then there will be glucose in the water of the cup because of diffusion from a high to a low concentration. Diffusion occurred when we put the starch and glucose dialysis bag into the water. When we first checked before we waited the 30 minutes there was no glucose present in the water. But after the 30 minutes was up there was glucose present. That is when diffusion happened. Glucose diffused into the water because it came from a high concentration to a low concentration.
Some things that were a struggle to us was the time management. We didn't really think it would have taken us a long time but we got done with the lab 5 minutes before our period was up. Also we didn't understand how to tie the string tightly so that took us a while too. Overall it was a successful experiment.
Analysis Questions:
1. What is simple diffusion according to the experiment performed?
Simple diffusion is the movement through a membrane from a high concentration to a low concentration.
2. What are 2 characteristics of substances that do not allow them to pass through the selectively permeable membrane of living cells?
The size and the charge of the substance do not allow them to pass through the selectively permeable membrane. If it is too large or there is a charge they will not allow them to pass through. Especially if the molecules are hydrophilic-it goes well with water.
3. What types of molecules more easily through living cell membranes?
Small molecules that don't have a charge move more easily through living cell membranes. Also hydrophobic molecules move more easily through a membrane. hydrophobic- does not react to water
4. Did the glucose molecules pass through the dialysis tubing? How do you know?
Yes the glucose did pass through the dialysis tubing because the test strip turned green therefore there was glucose molecules in the solution. When we first tested it the test strip did not test positive for glucose.
5. Did the starch molecules pass through the dialysis tubing? What evidence do you have to support this conclusion?
No because the solution with the iodine in it did not change to a blue color. Also the starch did not pass through because the solution did not change from the bronze color to the blue color.
6. Why did we use dialysis tubing as a model for a cell membrane?
Because it is also a semipermeable membrane. Therefore it only allows certain molecules to pass.
7.Why were the results for final starch and final glucose filled in already on Data table 1? (Hint: Would all of the solutes diffuse out of the dialysis bag? Why or why not?)
The results were already filled out on the chart because we knew that when diffusion occurs solutes move from a high to low concentration but they can't do that if they have too large of molecules.
In our lab it went pretty well. We saw diffusion in action. According to my hypothesis was correct; If diffusion occurs during the experiment then there will be glucose in the water of the cup because of diffusion from a high to a low concentration. Diffusion occurred when we put the starch and glucose dialysis bag into the water. When we first checked before we waited the 30 minutes there was no glucose present in the water. But after the 30 minutes was up there was glucose present. That is when diffusion happened. Glucose diffused into the water because it came from a high concentration to a low concentration.
Some things that were a struggle to us was the time management. We didn't really think it would have taken us a long time but we got done with the lab 5 minutes before our period was up. Also we didn't understand how to tie the string tightly so that took us a while too. Overall it was a successful experiment.
Analysis Questions:
1. What is simple diffusion according to the experiment performed?
Simple diffusion is the movement through a membrane from a high concentration to a low concentration.
2. What are 2 characteristics of substances that do not allow them to pass through the selectively permeable membrane of living cells?
The size and the charge of the substance do not allow them to pass through the selectively permeable membrane. If it is too large or there is a charge they will not allow them to pass through. Especially if the molecules are hydrophilic-it goes well with water.
3. What types of molecules more easily through living cell membranes?
Small molecules that don't have a charge move more easily through living cell membranes. Also hydrophobic molecules move more easily through a membrane. hydrophobic- does not react to water
4. Did the glucose molecules pass through the dialysis tubing? How do you know?
Yes the glucose did pass through the dialysis tubing because the test strip turned green therefore there was glucose molecules in the solution. When we first tested it the test strip did not test positive for glucose.
5. Did the starch molecules pass through the dialysis tubing? What evidence do you have to support this conclusion?
No because the solution with the iodine in it did not change to a blue color. Also the starch did not pass through because the solution did not change from the bronze color to the blue color.
6. Why did we use dialysis tubing as a model for a cell membrane?
Because it is also a semipermeable membrane. Therefore it only allows certain molecules to pass.
7.Why were the results for final starch and final glucose filled in already on Data table 1? (Hint: Would all of the solutes diffuse out of the dialysis bag? Why or why not?)
The results were already filled out on the chart because we knew that when diffusion occurs solutes move from a high to low concentration but they can't do that if they have too large of molecules.

Part 2:
Explanation: In this lab we looked at different amounts of sucrose solution in dialysis bags and weighed the initial mass and then waited a time period of 30 minutes and weighed the final mass.
Hypothesis: If 6 dialysis tubes were filled with different amounts of sucrose solution and placed in cups of water for 30 min, then after the time is up their mass will have increased due to osmosis.
Materials:
Explanation: In this lab we looked at different amounts of sucrose solution in dialysis bags and weighed the initial mass and then waited a time period of 30 minutes and weighed the final mass.
Hypothesis: If 6 dialysis tubes were filled with different amounts of sucrose solution and placed in cups of water for 30 min, then after the time is up their mass will have increased due to osmosis.
Materials:
- Goggles
- 6 cups
- 6 15-cm dialysis tubing segments
- 12 10-cm string segments
- 1 Medicine cup, plastic
- 10 ml Distilled water
- 10 ml 0.2 M Sucrose Solution
- 10 mL 0.4 M Sucrose Solution
- 10 mL 0.6 M Sucrose Solution
- 10 mL 0.8 M Sucrose Solution
- 10 mL 1.0 M Sucrose Solution
- 1 marking pencil
- Tape
- Paper towel
Procedures:
- Gently rub each piece of tubing between your fingers to open it.
- Tie one end of the tubing tightly with a piece of string. Fill the tubing with water and test it for leaks at a sink and then empty the tubing.
- Use the medicine cup to measure out 10 mL of the appropriate solution into the dialysis bag as follows:
- Rinse the outside of each bag under running water to remove excess sucrose solution present on the outside of the bag. Squeeze each bag gently to make sure there are no leaks. If there is a leak make sure you adjust the strings.
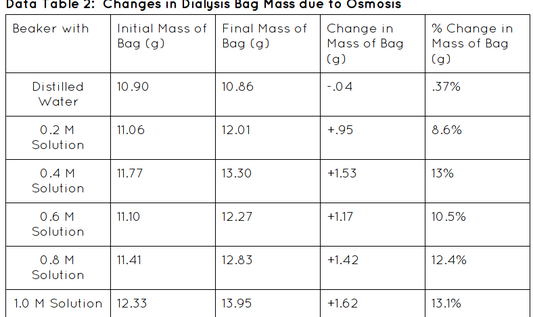
- Dry the outside of the bag with a paper towel and weigh the mass of each bag. Record in Data table 2.
- Place the appropriate dialysis bag into each of the numbered cups and fill each cup with enough distilled water to completely cover the bag. Begin recording the time. You need 30 min.
- After 30 min, remove each bag from its cup and gently blot excess moisture from the surface of each bag with a paper towel. Weigh the mass of each bag again. Record the final mass in Data table 2.
- Wash and clean up. Dispose of any waste materials.
- Calculate the change in mass for each bag. Record the information as accurately as possible.
- Divide the change in mass calculated in Step 9 by the initial mass of the appropriate bag and multiply by 100. This is the percent change in mass for each bag.
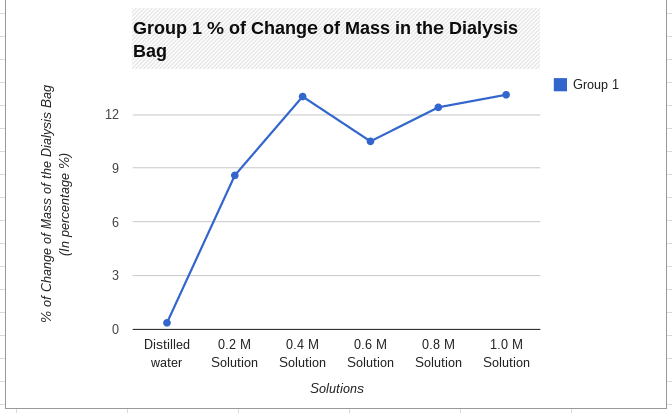
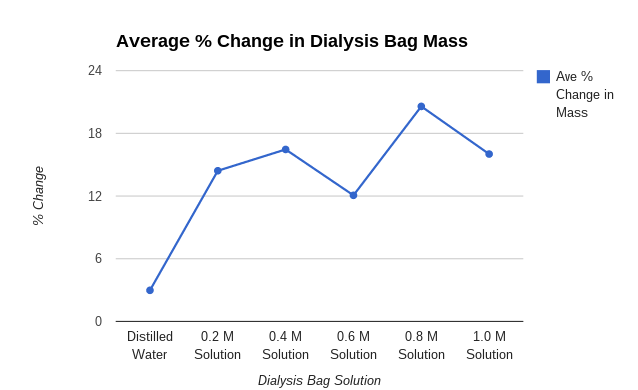
- Calculate the class average percent mass of the bags in Data table 2.
- Construct a graph of the percent change in mass for the average values and their own group’s values.
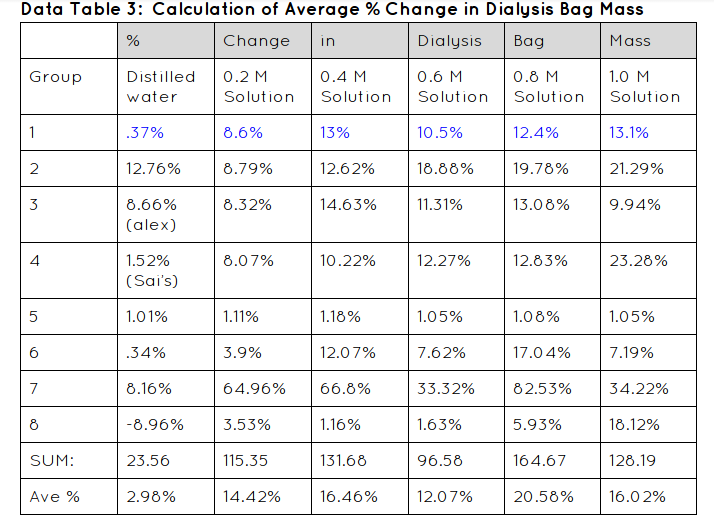
Graph:
Graph Justification:
I used a line graph because it worked with my data great. You can easily see the difference between the average and my group 1's data. You can tell that they are both accurate because they are in the same range. Using a line graph helps a lot because it helps you see the changes in data and it shows you if you were right or wrong with your data.
I used a line graph because it worked with my data great. You can easily see the difference between the average and my group 1's data. You can tell that they are both accurate because they are in the same range. Using a line graph helps a lot because it helps you see the changes in data and it shows you if you were right or wrong with your data.
Conclusion:
My hypothesis; If 6 dialysis tubes were filled with different amounts of sucrose solution and placed in cups of water for 30 min, then after the time is up their mass will have increased due to osmosis, was correct. It was correct because all of our bags' mass increased. None of them lost mass therefore we conducted a well put together lab.
In this experiment we conducted it very well but there were some struggles. We struggled with also time management and tying the dialysis bags. But overall it was a great experiment. When we first started we had a confusion but our teacher was super helpful and put us back in the right direction.
Analysis Questions:
1. What is osmosis according to the experiment performed? How does it differ from the diffusion we did in Part I?
According to the experiment performed, osmosis is the movement of water through a selectively permeable membrane from a region of higher concentration to a region of lower concentration. It differs from diffusion in the way that osmosis is only the movement of water molecules while diffusion is the movement of any molecules.
2. Did osmosis occur in each treatment? What observations led you to this conclusion?
Yes, osmosis occurred in each treatment. The observation that led me to this conclusion was the change in mass for each treatment, which means that the solutions went through the membrane and osmosis.
3. You observed a change in mass of the bags over the course of the experiment. Why was it important to convert the absolute change in mass of the bags to a percent change in mass of the bags?
Yes it is important to convert the mass of the bag into a percent because we would then be able to compare the different bags and keeps us from making any errors because we are dealing with a percent not over complicated numbers.
4. Did you see any clear relationship between the molarity of the solutions in the bags and the percent change in the mass of the bags? If so, how would you describe that relationship?
No I did not see a clear relationship between the molarity and the percent of change in the mass of the bags. The percent of change has to do with the weighing of the bag before and after osmosis occurred.
5. If you had placed all 6 bags into a beaker containing 1.2 M sucrose, would your results have different from what you observed? Why? Describe possible results for each of the bags placed in this solution.
Yes our results would have been different because our result would have turned out to be the complete same because we are putting the same sucrose solution inside them for the same amount of time. We would have no variation in the experiment.
My hypothesis; If 6 dialysis tubes were filled with different amounts of sucrose solution and placed in cups of water for 30 min, then after the time is up their mass will have increased due to osmosis, was correct. It was correct because all of our bags' mass increased. None of them lost mass therefore we conducted a well put together lab.
In this experiment we conducted it very well but there were some struggles. We struggled with also time management and tying the dialysis bags. But overall it was a great experiment. When we first started we had a confusion but our teacher was super helpful and put us back in the right direction.
Analysis Questions:
1. What is osmosis according to the experiment performed? How does it differ from the diffusion we did in Part I?
According to the experiment performed, osmosis is the movement of water through a selectively permeable membrane from a region of higher concentration to a region of lower concentration. It differs from diffusion in the way that osmosis is only the movement of water molecules while diffusion is the movement of any molecules.
2. Did osmosis occur in each treatment? What observations led you to this conclusion?
Yes, osmosis occurred in each treatment. The observation that led me to this conclusion was the change in mass for each treatment, which means that the solutions went through the membrane and osmosis.
3. You observed a change in mass of the bags over the course of the experiment. Why was it important to convert the absolute change in mass of the bags to a percent change in mass of the bags?
Yes it is important to convert the mass of the bag into a percent because we would then be able to compare the different bags and keeps us from making any errors because we are dealing with a percent not over complicated numbers.
4. Did you see any clear relationship between the molarity of the solutions in the bags and the percent change in the mass of the bags? If so, how would you describe that relationship?
No I did not see a clear relationship between the molarity and the percent of change in the mass of the bags. The percent of change has to do with the weighing of the bag before and after osmosis occurred.
5. If you had placed all 6 bags into a beaker containing 1.2 M sucrose, would your results have different from what you observed? Why? Describe possible results for each of the bags placed in this solution.
Yes our results would have been different because our result would have turned out to be the complete same because we are putting the same sucrose solution inside them for the same amount of time. We would have no variation in the experiment.
Part 3:
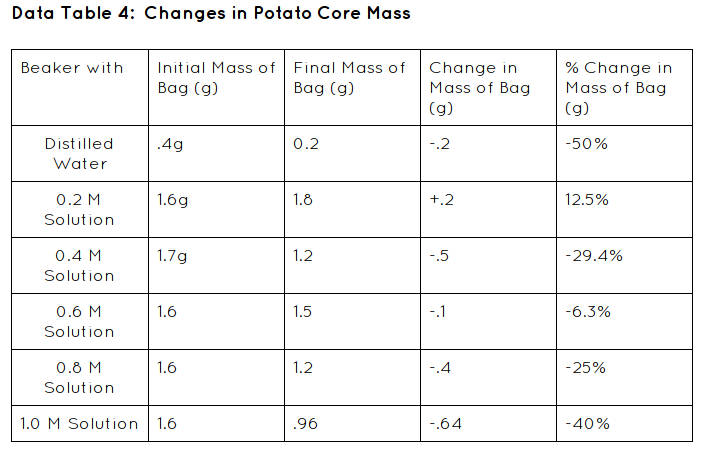
Explanation: In this experiment we were testing the rate of diffusion and osmosis. We are weighing potato cores for their initial mass and then we are waiting for a time period of 24 hours too check the final mass to see if they were to lose of gain weight from the sucrose solution.
Hypothesis:If the potatoes are hypotonic to the solution then they will lose mass.
Materials:
Procedures:
Explanation: In this experiment we were testing the rate of diffusion and osmosis. We are weighing potato cores for their initial mass and then we are waiting for a time period of 24 hours too check the final mass to see if they were to lose of gain weight from the sucrose solution.
Hypothesis:If the potatoes are hypotonic to the solution then they will lose mass.
Materials:
- Goggles
- #2 Cork borer
- 1 potato
- 1 scalpel
- 1 cm ruler
- 6 plastic cups
- plastic wrap or foil
- pencil
- 100 mL Distilled water
- 100 mL 0.2 Sucrose solution
- 100 mL 0.4 Sucrose solution
- 100 mL 0.6 Sucrose solution
- 100 mL 0.8 Sucrose solution
- 100 mL 1.0 Sucrose solution
- Paper towel
- Balance
Procedures:
- Push the cork borer completely through the potato. Remove the potato core from the borer. Carefully cut off each end of the core where the potato skin is.
- Lay the core next to a metric ruler. Measure and cut a 3 cm long potato core section.
- Punch a total of 4 potato cores, each 3 cm in length. Obtain the mass of the 4 cores together to the nearest 0.1 g. Keep cores covered until you are able to weight them.
- Place 4 cores into one of the labeled plastic cups containing a solution. Record the initial mass of the 4 cores for this solution in Data table 4.
- Repeat steps 1-5 with each of the remaining cups. Cover all of the cups with plastic wrap or foil to keep evaporation to a minimum.
- Place the cups in your tray so that they will not be disturbed overnight.
- Remove the cores from one of the cups and carefully place them on a paper towel. Dab the cores with the paper towel to dry them. Obtain the mass of the 4 cores together. Record in Data table 4.
- Repeat step 7 for each of the cups.
- For each of te solutions, subtract the initial mass of the cores from the final mass of the cores to obtain the change in mass. Be sure to record a positive result if the cores gained mass and a negative result if the cores lost mass. Record these calculations in Data Table 4.
- For each solution, calculate the percent change in mass for the cores as you did for the dialysis bags in the previous exercise. Record.
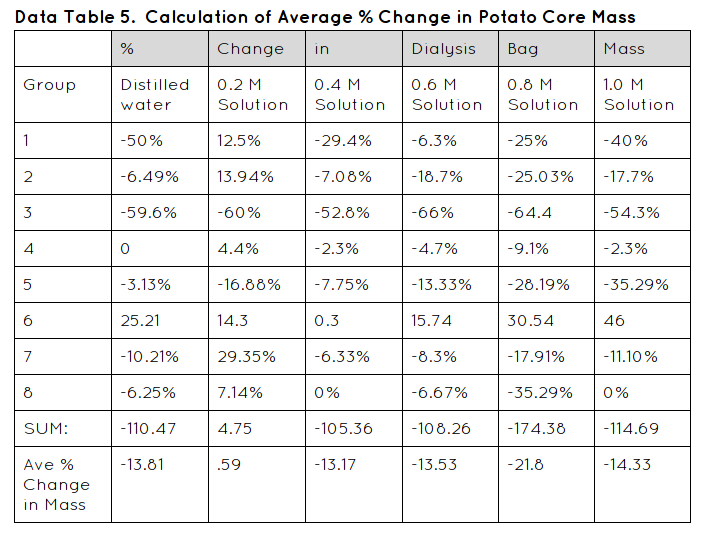
- Calculate the class average percent change in mass of the cores in each solution in Data table 5.
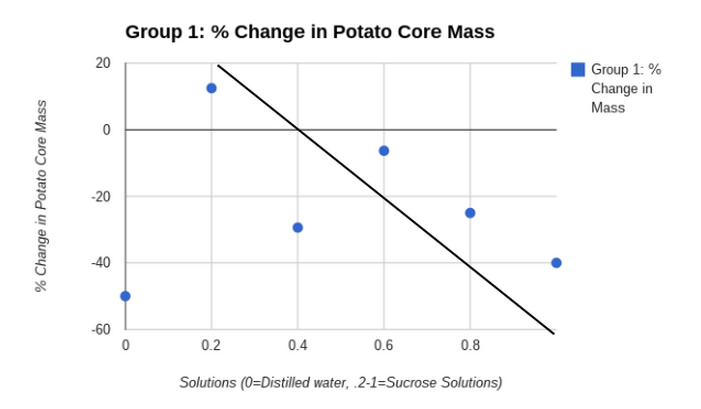
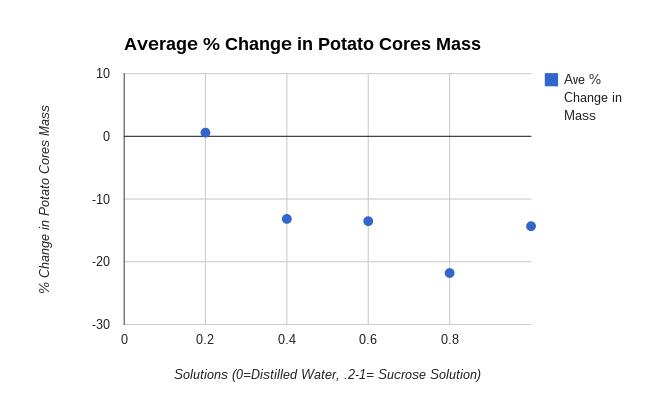
- Construct a graph of the percent change in mass for both your group’s data and the combined class averages.
- Draw a “best fit” line corresponding to the points on your graph. The point at which your line crosses zero on the Y-axis is an approximation of the molar concentration of solutes inside the potato tuber cells. This point identifies the molarity of a sucrose solution that has the same water potential as that of the potato tuber cells.
Graphs:
Graph Justification:
I used a scattered plot graph because the data was easily seen, it also organized my data very well. The data was easy to compare to my other scattered plot graph and we could create a line of best fit to the points. The line of best fit was needed in our analysis questions so that is another reason that we needed to have a scattered plot graph.
Conclusion:
In my lab my data was supported. The initial mass decreased over the time period of 24 hours because the weight of the potato cores all mostly lost weight except for one in the .2 sucrose solution. Therefore my hypothesis was not correct; If the potatoes are hypotonic to the solution then they will lose mass, because not all of my potato cores lost weight.
Some errors in my lab might have been because we didn't put the potato cores in the solutions all at the same time so it might have messed up my data a little bit. Another error might have been when another group dumped their solutions out by mistake so we gave them some of our sucrose solutions so they could conduct their experiment. It probably messed with our data because we needed all the solution and we didn't have any. Another data that could have messed with my data was not fully covering the potatoes. It could have lead to an error of the solution and the potatoes soaking up the solution.
Analysis Questions:
1. Calculate the solute potential for the sucrose solution.
.5 M
2. Calculate the Potato tuber cell water potential.
-12.38 bars
3. Why was it necessary to keep the potato tuber cores in a covered container until the balance was available? If you had left them out in the open, how might that have affected your results?
It was necessary to cover the container because the water would eventually start to evaporate. If the water evaporated the concentration would become greater because there would be the same amount of solute but less solvent. It would then mess up our lab because this would cause more water to diffuse into the potato cores.
4. Some plants grow in salt marshes, where the concentration of solutes in the soil water is high. What is one adaptation you might expect to find in these salt-tolerant plants with respect to the solute concentration within their cells?
One adaption we might find is that they have a high solute concentration. If they didn't have this adaption then they wouldn't be able to survive because the salty water would absorb all the water and it would result in the plants dying because they wouldn't be getting any oxygen and nutrients from the water. They are able to take on all of the salt water because they have cells that already have enough salt because they have a high solute concentrations so they take in the water because it is more needed.
5. Water is usually “pulled” through a plant, from roots to leaves. What happens at the leaves to cause this “pull”? How would you describe the water potential at the leaf versus the water potential at the root? Why?
To cause this pull transpiration needs to occur. It causes the stomata (pores on the leaves surfaces) to breathe in the carbon dioxide and (water) and to release the oxygen through the process. At the leaves water gets evaporated and it causes a pull towards the leaves in order to fill the holes where the evaporated water was. At the root the water is sucked in due to water potential. It determines the movement and direction of water. It is going to move from a higher water potential to a lower water potential.
I used a scattered plot graph because the data was easily seen, it also organized my data very well. The data was easy to compare to my other scattered plot graph and we could create a line of best fit to the points. The line of best fit was needed in our analysis questions so that is another reason that we needed to have a scattered plot graph.
Conclusion:
In my lab my data was supported. The initial mass decreased over the time period of 24 hours because the weight of the potato cores all mostly lost weight except for one in the .2 sucrose solution. Therefore my hypothesis was not correct; If the potatoes are hypotonic to the solution then they will lose mass, because not all of my potato cores lost weight.
Some errors in my lab might have been because we didn't put the potato cores in the solutions all at the same time so it might have messed up my data a little bit. Another error might have been when another group dumped their solutions out by mistake so we gave them some of our sucrose solutions so they could conduct their experiment. It probably messed with our data because we needed all the solution and we didn't have any. Another data that could have messed with my data was not fully covering the potatoes. It could have lead to an error of the solution and the potatoes soaking up the solution.
Analysis Questions:
1. Calculate the solute potential for the sucrose solution.
.5 M
2. Calculate the Potato tuber cell water potential.
-12.38 bars
3. Why was it necessary to keep the potato tuber cores in a covered container until the balance was available? If you had left them out in the open, how might that have affected your results?
It was necessary to cover the container because the water would eventually start to evaporate. If the water evaporated the concentration would become greater because there would be the same amount of solute but less solvent. It would then mess up our lab because this would cause more water to diffuse into the potato cores.
4. Some plants grow in salt marshes, where the concentration of solutes in the soil water is high. What is one adaptation you might expect to find in these salt-tolerant plants with respect to the solute concentration within their cells?
One adaption we might find is that they have a high solute concentration. If they didn't have this adaption then they wouldn't be able to survive because the salty water would absorb all the water and it would result in the plants dying because they wouldn't be getting any oxygen and nutrients from the water. They are able to take on all of the salt water because they have cells that already have enough salt because they have a high solute concentrations so they take in the water because it is more needed.
5. Water is usually “pulled” through a plant, from roots to leaves. What happens at the leaves to cause this “pull”? How would you describe the water potential at the leaf versus the water potential at the root? Why?
To cause this pull transpiration needs to occur. It causes the stomata (pores on the leaves surfaces) to breathe in the carbon dioxide and (water) and to release the oxygen through the process. At the leaves water gets evaporated and it causes a pull towards the leaves in order to fill the holes where the evaporated water was. At the root the water is sucked in due to water potential. It determines the movement and direction of water. It is going to move from a higher water potential to a lower water potential.




 RSS Feed
RSS Feed
